Structural basis of phage terminase recognition by the bacterial immune receptor Avs2
Chiu, C., Evans, S.A., Wilkinson, M.E., Li, D., Zhang, F., Gao, A.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| AVAST type 2 anti-phage system protein Avs2 | 1,484 | Escherichia coli | Mutation(s): 0 | 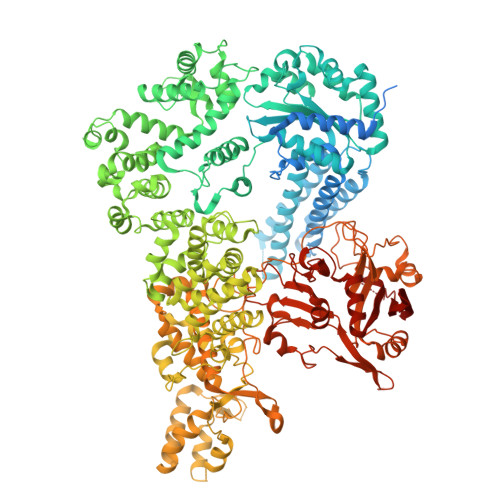 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Terminase, large subunit | 586 | Escherichia phage PhiV-1 | Mutation(s): 0 EC: 3.6.4 (PDB Primary Data), 3.1.21 (PDB Primary Data) | 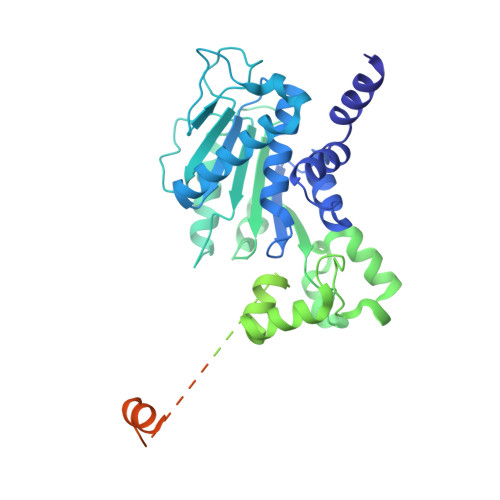 | |
UniProt | |||||
Find proteins for A0A7G3WWS0 (Escherichia phage PhiV-1) Explore A0A7G3WWS0 Go to UniProtKB: A0A7G3WWS0 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A7G3WWS0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ATP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | CA [auth E] EA [auth F] GA [auth G] I [auth A] IA [auth H] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| MG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AA [auth D] BA [auth D] DA [auth E] FA [auth F] HA [auth G] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | RELION | 4.0 |
| MODEL REFINEMENT | Coot | |
| MODEL REFINEMENT | ISOLDE | |
| MODEL REFINEMENT | PHENIX |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Other private | United States | -- |