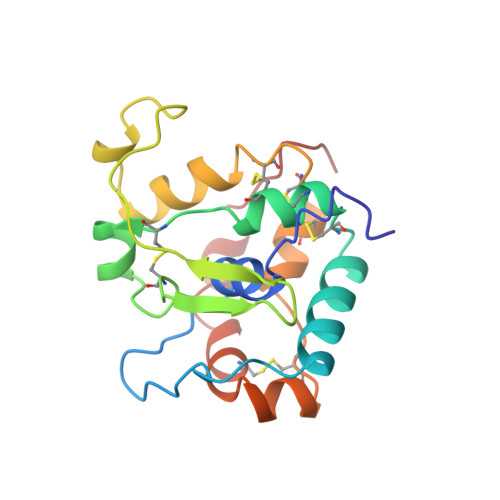
Hidden Structural States of Proteins Revealed by Conformer Selection
Huang, Y.J., Ramelot, T.A., Spaman, L.E., Kobayashi, N., Montelione, G.T.(2026) Nat Commun
- PubMed: 42045216 Search on PubMed
- DOI: https://doi.org/10.1038/s41467-026-71997-6
- Primary Citation Related Structures:
9A8V, 9A8Z - PubMed Abstract:
We introduce AISAR (AI SAmpling with NMR Recall selection), a computational-experimental framework for identifying alternative conformational states from NMR data. Unlike conventional NMR methods that rely on spatial restraints, AISAR combines AI-driven conformational sampling of realistic models with Bayesian-like scoring against NOESY and other NMR observables. Applied to Gaussia luciferase, AISAR reveals two interconverting states involving large rearrangements of two lids, binding pockets, and cryptic surface cavities. AISAR also identifies two distinct conformational states of the human tumor suppressor Cyclin-Dependent Kinase 2-Associated Protein 1, demonstrating its utility across diverse protein scaffolds. Validation using the NOESY Double Recall method shows that these multistate models account for NOESY peaks that are not explained by single-state models, supporting the presence of fast-exchanging structural states in dynamic equilibrium. AISAR enables detection and evaluation of conformational heterogeneity and cryptic pockets not resolved by conventional single-state NMR analysis, providing insights into protein structural dynamics and function.
- Department of Chemistry and Chemical Biology, Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, New York, 12180, USA. huangy26@rpi.edu.
Organizational Affiliation: